Clinical Trial Data Management Using SAS (2026 Complete Guide)
Clinical Trial Data Management Using SAS is a critical function in modern clinical research that ensures the accuracy, integrity, traceability, and regulatory compliance of clinical trial data. In 2026, with global trials becoming more decentralized and data-intensive, the demand for structured data management using SAS and CDISC standards has significantly increased.
Introduction to Clinical Trial Data Management
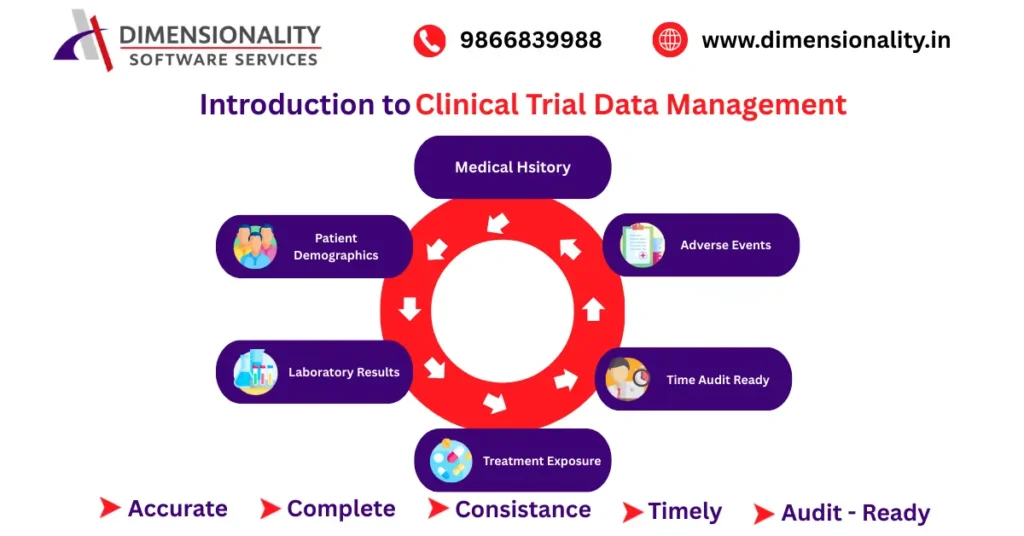
Clinical Trial Data Management Using SAS is a critical function in modern clinical research that ensures the accuracy, integrity, traceability, and regulatory compliance of clinical trial data. In 2026, with global trials becoming more decentralized and data-intensive, the demand for structured data management using SAS and CDISC standards has significantly increased.
Clinical trials are structured investigations designed to evaluate the safety and efficacy of investigational medicinal products. These studies generate large volumes of structured and semi-structured data including
- Patient demographics
- Medical history
- Adverse events
- Laboratory results
- Concomitant medications
- Vital signs
Clinical Trial Data Management ensures that this data is
- Accurate
- Complete
- Consistent
- Timely
- Audit-ready
Without effective CTDM, statistical analysis may produce unreliable results, and regulatory approvals may be delayed or rejected.
Objectives of Clinical Trial Data Management
Without effective CTDM, statistical analysis may produce unreliable results, and regulatory approvals may be delayed or rejected.
The primary objectives of CTDM include
- Data Accuracy
Ensuring all collected data reflects true subject information.
- Data Completeness
Minimizing missing data and resolving discrepancies.
- Data Consistency
Maintaining logical alignment across datasets.
- Timely Database Lock
Delivering clean datasets within project timelines.
- Regulatory Compliance
Meeting global standards such as
U.S. Food and Drug Administration
European Medicines Agency
CDISC
ICH
In 2026, regulatory scrutiny is stronger than ever, making traceability and documentation mandatory.
Sources of Clinical Trial Data

Clinical trial data originates from multiple integrated systems. Effective data reconciliation is a key responsibility of the data management team.
Major Data Sources
Electronic Data Capture (EDC) – Case Report Form (CRF) data
Central Laboratories – Lab test results
ECG & Imaging Vendors – Specialized diagnostics
IWRS/IVRS – Randomization and drug supply
PRO/ePRO – Patient-reported outcomes
Modern trials often involve decentralized and hybrid designs, increasing the complexity of data integration.
Clinical Trial Data Management Lifecycle
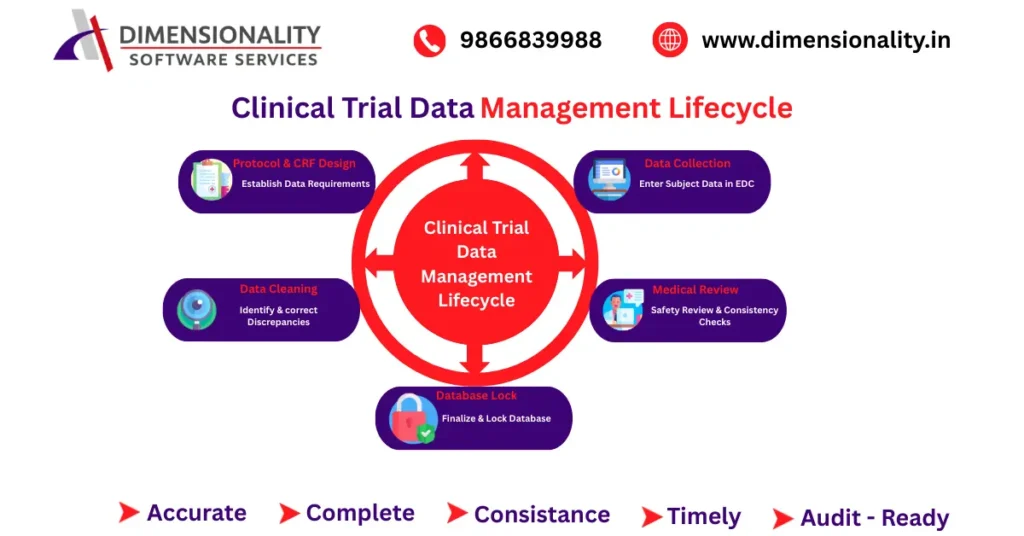
The CTDM lifecycle spans from protocol development to database lock.
Protocol & CRF Design
Data requirements are defined based on study objectives. Proper CRF design reduces downstream errors.
Data Collection
Clinical sites enter subject data into EDC systems.
Data Cleaning
Validation checks identify
Missing values
Out-of-range data
Logical inconsistencies
Protocol deviations
Medical Review
Clinical experts review safety signals and consistency.
Database Lock
All queries are resolved, and the dataset is frozen for analysis.
A clean database ensures smooth statistical programming and regulatory submission.
Role of SAS in Clinical Trial Data Management
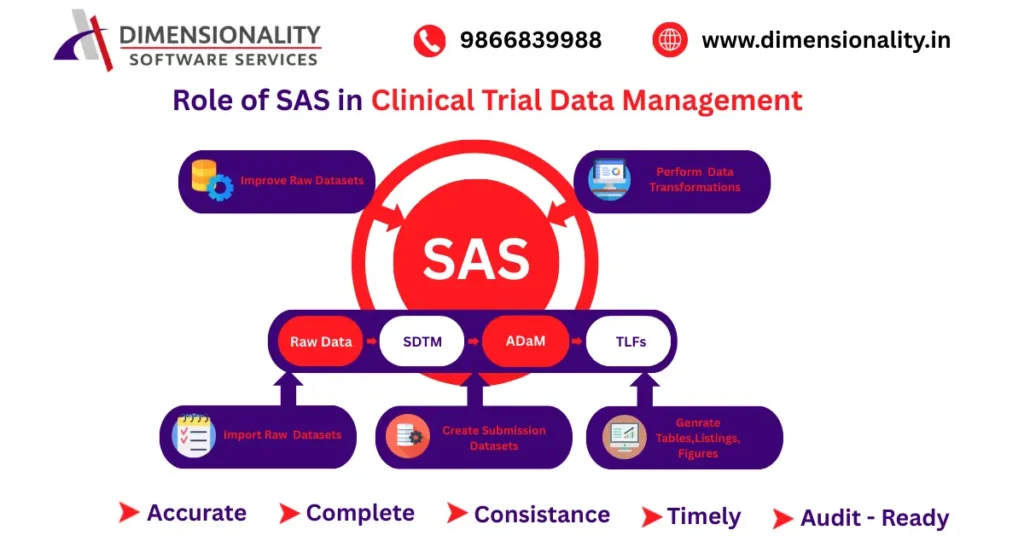
SAS Institute provides one of the most widely accepted tools in regulatory environments.
Why SAS Is Preferred in Clinical Trials
Regulatory acceptance
Robust data handling
Strong audit trail capabilities
Compatibility with CDISC standards
High-performance data processing
SAS Workflow in Clinical Data
Raw Data → SDTM → ADaM → TLFs
SAS is used to
Import raw datasets
Perform data transformations
Execute validation checks
Create standardized submission datasets
Generate Tables, Listings, and Figures (TLFs)
Because regulatory agencies rely heavily on SAS datasets, proficiency in SAS remains highly valuable in 2026
Study Data Tabulation Model (SDTM)

Study Data Tabulation Model is a CDISC standard that structures clinical data for submission.
Common SDTM Domains
DM – Demographics
AE – Adverse Events
LB – Laboratory Tests
VS – Vital Signs
EX – Exposure
Standardization ensures that reviewers at the FDA and EMA can navigate datasets efficiently.
Benefits of SDTM
Consistent structure
Faster regulatory review
Improved traceability
Analysis Data Model (ADaM)

Analysis Data Model supports statistical analysis.
ADaM datasets are derived from SDTM and include:
Derived variables
Analysis flags
Treatment group indicators
Time-to-event parameters
Core Dataset: ADSL
ADSL contains one record per subject and forms the backbone of all analysis datasets.
ADaM ensures:
Traceability
Reproducibility
Statistical transparency
Data Traceability in Clinical Trials
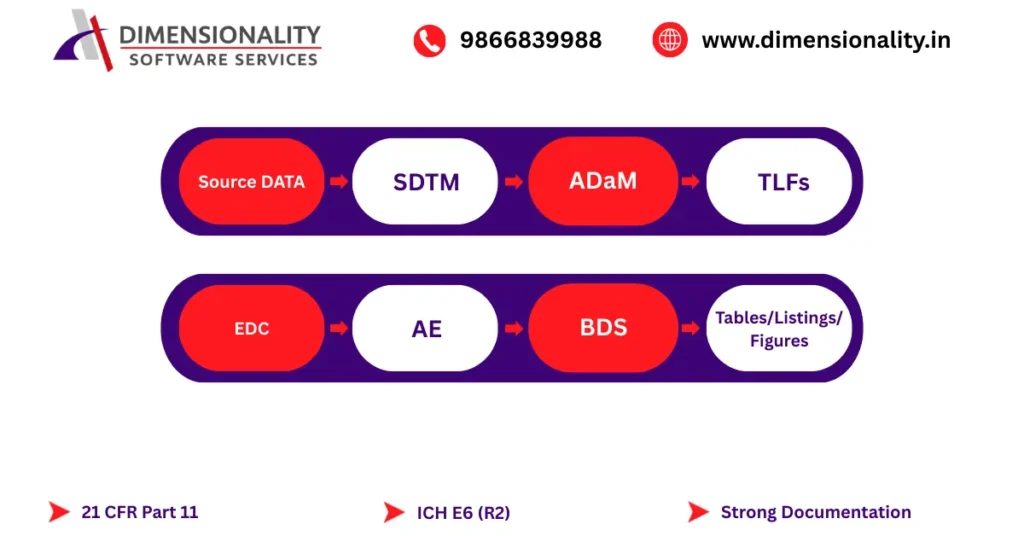
Traceability is a regulatory requirement in 2026.
Every value in a table must trace back to its source
Source Data → SDTM → ADaM → TLF
This ensures audit readiness and regulatory confidence
Traceability supports compliance with
21 CFR Part 11
ICH E6(R2)
Strong documentation is no longer optional—it is mandatory.
Quality Control (QC) in Clinical Data Management
Quality Control minimizes submission risks.
QC Activities Include
Double programming
Independent dataset review
Cross-dataset reconciliation
TLF validation
Documentation review
Effective QC reduces costly rework during regulatory submission.
Regulatory Guidelines and Compliance
Clinical Trial Data Management must comply with global standards
ICH Good Clinical Practice (GCP)
Ensures ethical and scientific quality.
FDA 21 CFR Part 11
Governs electronic records and signatures.
CDISC Standards (SDTM & ADaM)
Required for electronic submissions.
Failure to comply may result in submission rejection or delay.
Career Scope in Clinical Trial Data Management Using SAS (2026 Outlook)
The pharmaceutical and biotech industries are expanding rapidly in India and globally.
Common roles include
Clinical Data Analyst
SAS Programmer
SDTM Programmer
ADaM Programmer
Clinical Data Manager
Skills required
SAS programming
CDISC standards
Understanding of clinical protocols
Data validation techniques
With increasing regulatory requirements, professionals skilled in SAS-based clinical data management remain in high demand.
Future Trends in 2026
Clinical data management is evolving with
Risk-based monitoring
AI-assisted data cleaning
Real-time data integration
Decentralized clinical trials
Cloud-based data platforms
However, despite automation growth, SAS and CDISC standards remain foundational for regulatory submissions.
Best Practices for Clinical Trial Data Management Using SAS
- Design CRFs carefully
- Implement early validation checks
- Maintain detailed documentation
- Ensure SDTM compliance from start
- Perform independent QC
- Maintain strong traceability
- Stay updated with regulatory guidelines
Adopting these practices improves efficiency and submission success rates.
Conclusion
Clinical Trial Data Management Using SAS is an essential discipline that ensures data integrity, regulatory compliance, and scientific validity in clinical research. From raw data collection to SDTM and ADaM dataset preparation, SAS plays a pivotal role in transforming clinical data into submission-ready formats.
In 2026, as global regulations become stricter and clinical trials become more complex, structured data management using SAS and CDISC standards remains the backbone of successful regulatory submissions.
For students and professionals entering clinical research, mastering Clinical Trial Data Management Using SAS opens strong career opportunities in pharmaceutical companies, CROs, and global research organizations.
