Clinical Trial Data Management Using SAS: Complete Guide to Lifecycle, SDTM, ADaM and Data Validation
Introduction to Clinical Trial Data Management Using SAS

Clinical trials are essential for evaluating the safety and effectiveness of new drugs, medical devices, and treatment methods. These studies generate massive amounts of data from multiple sources such as hospitals, laboratories, and patient reports. Managing this data accurately is extremely important for the success of any clinical study.
Clinical Trial Data Management (CTDM) refers to the process of collecting, validating, cleaning, and organizing clinical trial data so it can be analyzed and submitted to regulatory authorities. The quality of data management directly impacts the reliability of clinical trial results.
Today, most pharmaceutical companies and clinical research organizations rely on SAS (Statistical Analysis System) to manage and analyze clinical trial data efficiently. SAS plays a key role in transforming raw clinical data into standardized datasets that can be used for statistical analysis and regulatory submissions.
If you want to learn these concepts in a structured way, you can explore our Clinical SAS Training Program in Hyderabad
In this article, we will explore clinical trial data management using SAS, including the lifecycle, data sources, validation processes, and the importance of SDTM and ADaM datasets in clinical research.
What is Clinical Trial Data Management?
Clinical Trial Data Management is a specialized discipline within clinical research that focuses on ensuring high-quality, accurate, and reliable data throughout the entire clinical trial process.
The primary goals of clinical data management include
- Ensuring data accuracy and completeness
- Maintaining data consistency across multiple systems
- Protecting patient confidentiality
- Ensuring compliance with regulatory guidelines
- Delivering clean datasets for statistical analysis
Clinical trials generate different types of data such as
- Patient demographic information
- Medical history
- Adverse events
- Laboratory test results
- Vital signs
- Drug exposure details
Without proper data management, it becomes difficult to draw valid conclusions about the safety and effectiveness of a drug.
If you are new to clinical programming, you can first understand Clinical SAS Programming basics here
Range Checks
Verify that numerical values fall within acceptable ranges.
Logical Consistency Checks
Ensure that related data fields are logically consistent.
Protocol Deviation Checks
Identify data that does not follow the study protocol.
SAS programs are used to automate many of these validation processes, helping data managers quickly identify and resolve data issues.
When errors are detected, queries are sent to clinical trial sites for clarification or correction.
Role of SAS in Clinical Trial Data Management

SAS is one of the most widely used tools in the pharmaceutical and clinical research industry. It helps researchers manage, process, and analyze large volumes of clinical data efficiently.
SAS is used in several stages of clinical trial data management, including
- Importing raw clinical data from multiple sources
- Performing data cleaning and validation checks
- Transforming raw data into standardized formats
- Creating analysis-ready datasets
- Generating statistical reports and outputs
The typical data flow in clinical trials looks like this:
Raw Data → SDTM → ADaM → TLF (Tables, Listings, Figures)
SAS programmers play a crucial role in converting raw clinical trial data into standardized datasets that meet regulatory requirements.
Clinical Trial Data Management Lifecycle
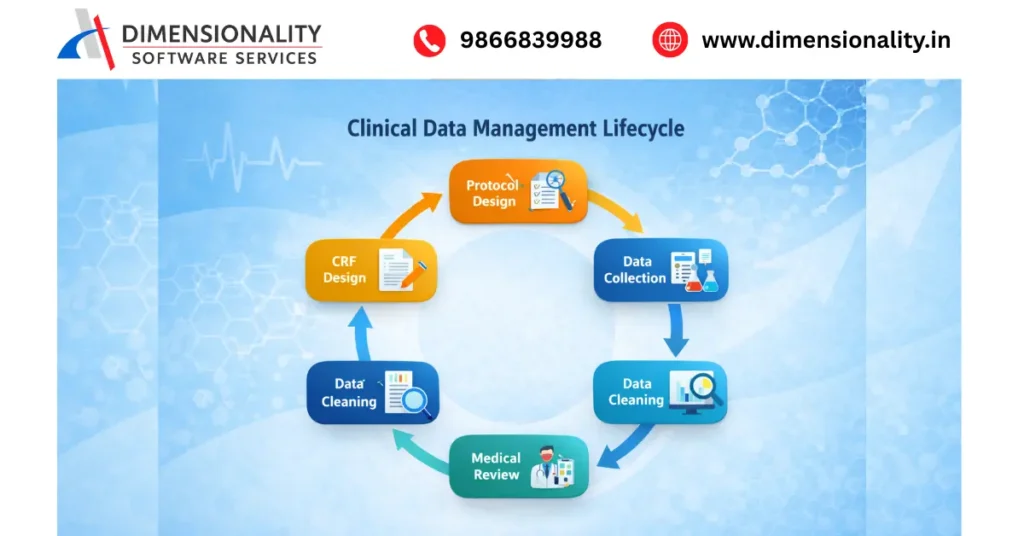
The clinical data management lifecycle consists of several stages that ensure the quality and integrity of clinical trial data.
Protocol Design
The clinical trial protocol defines the objectives, methodology, and data requirements of the study.
CRF Design (Case Report Form)
Case Report Forms are designed to collect patient data during the trial.
Data Collection
Data is collected from clinical trial sites using systems such as Electronic Data Capture (EDC).
Data Cleaning
The collected data is reviewed to identify errors, missing values, or inconsistencies.
Medical Review
Medical experts review the data to ensure clinical consistency.
Medical Review
Once all queries are resolved, the database is locked and prepared for statistical analysis.
Sources of Clinical Trial Data

Clinical trial data comes from multiple systems and vendors. Data managers must integrate and manage this information effectively.
Common data sources include
Electronic Data Capture (EDC)
Used to collect patient data electronically at clinical trial sites.
Laboratory Data
Central laboratories provide results for blood tests and other diagnostic tests.
ECG and Imaging Vendors
Provide specialized diagnostic data such as heart monitoring or imaging results.
IWRS (Interactive Web Response System)
Used for randomization and drug supply management.
PRO (Patient Reported Outcomes)
Patients provide information about their symptoms and quality of life.
Managing data from these different sources requires strong data integration and validation processes.
For more information about clinical data standards used in clinical trials, you can visit the CDISC official website
Data Cleaning and Validation Using SAS

Data cleaning is a critical step in clinical trial data management. It ensures that the data used for analysis is accurate and reliable.
Common data validation checks include
Missing Data Checks
Identify missing values in important fields.
Understanding SDTM in Clinical SAS
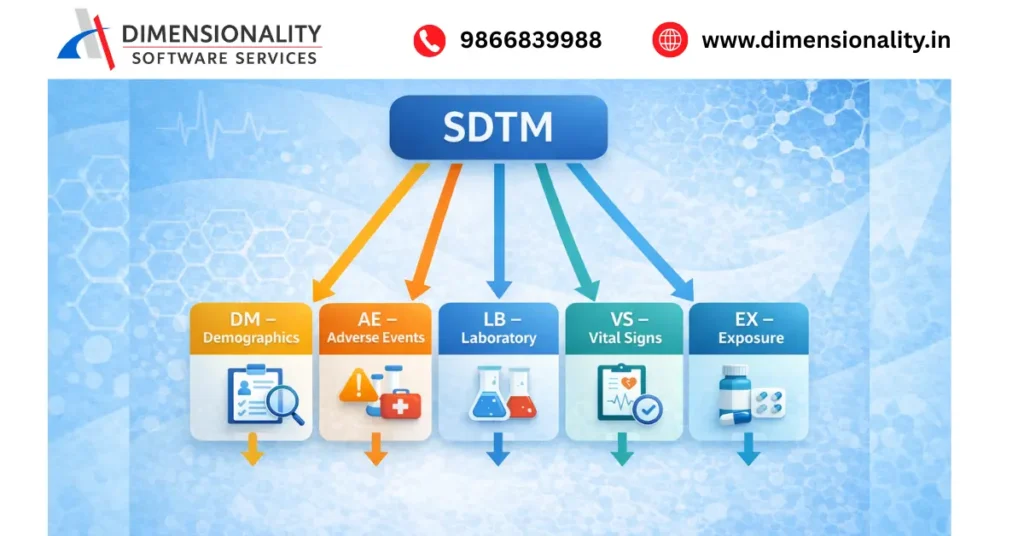
SDTM stands for Study Data Tabulation Model, which is a data standard developed by CDISC (Clinical Data Interchange Standards Consortium).
SDTM provides a standardized structure for organizing clinical trial data before submitting it to regulatory agencies such as the FDA.
SDTM datasets are organized into different domains, including
- DM – Demographics
- AE – Adverse Events
- LB – Laboratory Data
- VS – Vital Signs
- EX – Exposure
Using standardized SDTM datasets allows regulatory reviewers to understand clinical trial data quickly and efficiently.
Understanding ADaM in Clinical SAS

ADaM stands for Analysis Data Model. These datasets are derived from SDTM datasets and are specifically designed for statistical analysis.
ADaM datasets include derived variables, analysis flags, and parameters required for statistical calculations.
One of the most important ADaM datasets is
ADSL (Subject Level Dataset)
This dataset contains one record per subject and serves as the foundation for many statistical analyses.
ADaM datasets ensure that all statistical results can be reproduced and traced back to the original data source.
Data Traceability in Clinical Trials

Data traceability is a critical regulatory requirement in clinical research. It ensures that every analysis result can be traced back to the original source data.
The traceability path typically follows this structure
Source Data → SDTM → ADaM → TLF
This traceability ensures transparency and builds confidence in clinical trial results during regulatory review.
Quality Control in Clinical Data Management
Quality Control (QC) processes help ensure that clinical trial datasets and analysis outputs are accurate.
Common QC activities include
- Double programming validation
- Independent dataset review
- Data reconciliation across sources
- Verification of tables, listings, and figures
Regulatory Guidelines in Clinical Trials
Clinical trial data management must comply with strict regulatory guidelines to ensure data integrity and patient safety.
Some important regulatory standards include
ICH Good Clinical Practice (GCP)
Defines international standards for designing and conducting clinical trials.
FDA 21 CFR Part 11
Regulates electronic records and electronic signatures.
CDISC Standards
Includes SDTM and ADaM data standards used for regulatory submissions.
Compliance with these regulations is essential for gaining regulatory approval for new drugs.
Career Opportunities in Clinical SAS
Clinical SAS is one of the fastest-growing career fields in the pharmaceutical industry.
Common job roles include
- Clinical SAS Programmer
- Statistical Programmer
- Clinical Data Analyst
- Clinical Data Manager
Professionals with skills in SAS programming, SDTM, and ADaM are in high demand across pharmaceutical companies and clinical research organizations.
Learn Clinical Trial Data Management Using SAS
If you are interested in building a career in the pharmaceutical and clinical research industry, learning SAS programming and clinical data standards is an excellent starting point.
Our Clinical SAS Training in Hyderabad program is designed to help students and professionals gain practical knowledge of clinical trial data management, SDTM, ADaM datasets, and SAS programming used in real-world clinical studies.
This training helps learners develop the skills required to work as a Clinical SAS Programmer or Clinical Data Analyst in the clinical research industry.
Conclusion
Clinical Trial Data Management plays a critical role in ensuring the reliability and integrity of clinical research data. With the increasing complexity of clinical trials, effective data management processes have become more important than ever.
SAS remains one of the most powerful tools used in clinical research for managing, validating, and analyzing clinical trial data. By using standardized models such as SDTM and ADaM, organizations can ensure regulatory compliance and produce high-quality datasets for statistical analysis.
Understanding clinical trial data management using SAS provides valuable knowledge for anyone interested in pursuing a career in the clinical research or pharmaceutical industry.
