Introduction to SAS Programming for Clinical Studies
Introduction to SAS Programming for Clinical Studies

The healthcare and pharmaceutical industries are rapidly growing, creating huge demand for professionals skilled in clinical data analysis. One of the most in-demand skills today is Clinical SAS programming.
If you are looking to build a career in this field, enrolling in the clinical sas training in hyderabad can help you gain the right knowledge, practical skills, and job opportunities.
What is Clinical SAS?

Clinical SAS refers to the use of SAS software in managing, analyzing, and reporting clinical trial data. It plays a major role in ensuring the safety and effectiveness of medical treatments.
Clinical SAS is widely used for
- Data Management
- Statistical Analysis
- Safety Reporting
- SDTM & ADaM Dataset Creation
- Clinical Report Generation
- Automation using SAS Macros
These functions make it a core part of clinical data management.
What is a Clinical trial?

A clinical trial is a scientific study conducted to test new
- Medicines
- Medical devices
- Treatment procedures
The goal is to ensure safety, effectiveness, and reliability before releasing them to the public.
Phases of Clinical Trials
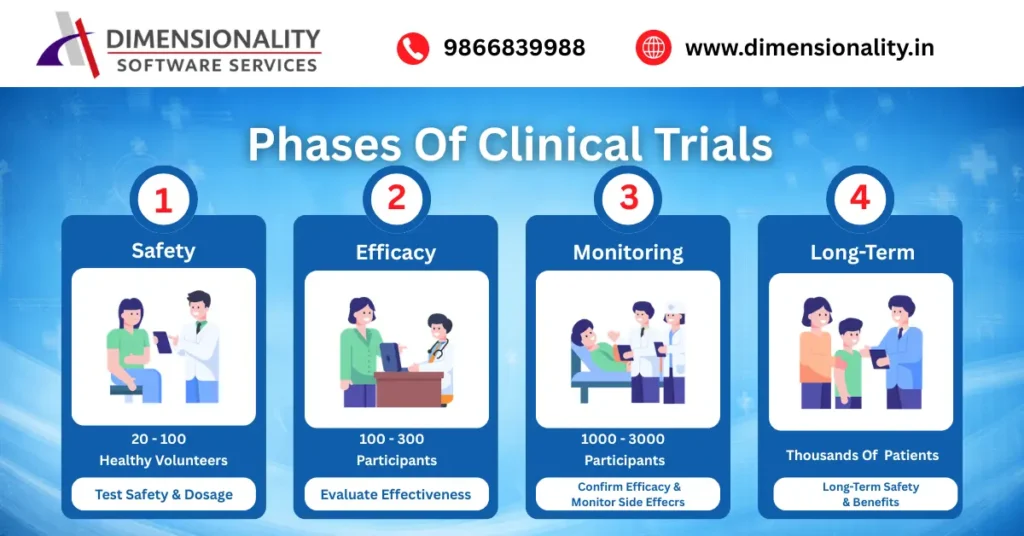
Clinical trials are conducted in multiple phases
Phase 0 – Microdosing
- Small group (10–15 participants)
- Focus: Drug behavior in the body
Phase 1 – Safety Testing
- 20–100 healthy volunteers
- Focus: Safety and dosage
Phase 2 – Effectiveness
- 100–300 patients
- Focus: Effectiveness and side effects
Phase 3 – Confirmation
- Large population (1000+)
- Focus: Safety and effectiveness
Phase 4 – Post-Marketing
- Real-world usage
- Focus: Long-term safety
Each phase ensures proper validation of treatments.
Clinical Trial Study Design

Clinical trials use different designs to reduce bias and improve accuracy
- Randomized Controlled Trials (RCT)
- Double-Blind Studies
- Crossover Trials
- Parallel Group Trials
- Single-Arm Trials
These designs are essential in clinical research jobs and real-time project work.
Understanding CDISC Standards

CDISC standards are used globally to standardize clinical trial data.
Key Standards
- SDTM (Study Data Tabulation Model) → Raw data structure
- ADaM (Analysis Data Model) → Analysis datasets
- Define-XML → Metadata for regulatory submission
Learning these standards is a key part of SDTM ADaM training.
Difference Between SDTM and ADaM
| Feature | SDTM | ADaM |
| Purpose | Data Standardization | Data Analysis |
| Usage | Regulatory Submission | Statistical Reporting |
| Data Type | Raw Structured Data | Derived datasets |
Clinical SAS Workflow

The complete workflow in clinical SAS includes
- Data collection using CRF
- Conversion of raw data into SDTM
- Creation of ADaM datasets
- Generation of Tables, Figures, Listings (TFLs)
- Final reporting for regulatory submission
This workflow is a must-learn in any SAS programming course.
Why Choose Clinical SAS as a Career?

Here are some strong reasons
- High demand in pharmaceutical companies
- Excellent salary growth
- Opportunities in global companies
- Strong career in clinical research jobs
- Work on real-time healthcare data
Because of these benefits, many students are now choosing clinical SAS training in hyderabad to start their careers.
Start Your Career with Clinical SAS Training in Hyderabad

To succeed in this field, proper training is very important. A good training program will provide
- Strong foundation in SAS programming
- Hands-on experience with SDTM & ADaM
- Real-time project exposure
- Interview preparation
- Placement assistance
Conclusion
Clinical SAS programming is one of the most powerful and in-demand skills in the healthcare and pharmaceutical industries today. Professionals with knowledge of SAS programming, SDTM, ADaM datasets, clinical trial processes, and reporting standards are highly valued across global clinical research organizations. Learning these skills opens doors to strong career opportunities, attractive salaries, and long-term growth in the pharma and biotechnology sectors.
If you are planning to enter the clinical research field, this is the right time to join Clinical SAS Training in Hyderabad and gain hands-on experience with real-time projects, certification support, and industry-relevant skills to build a successful and future-ready career in clinical data analysis.
